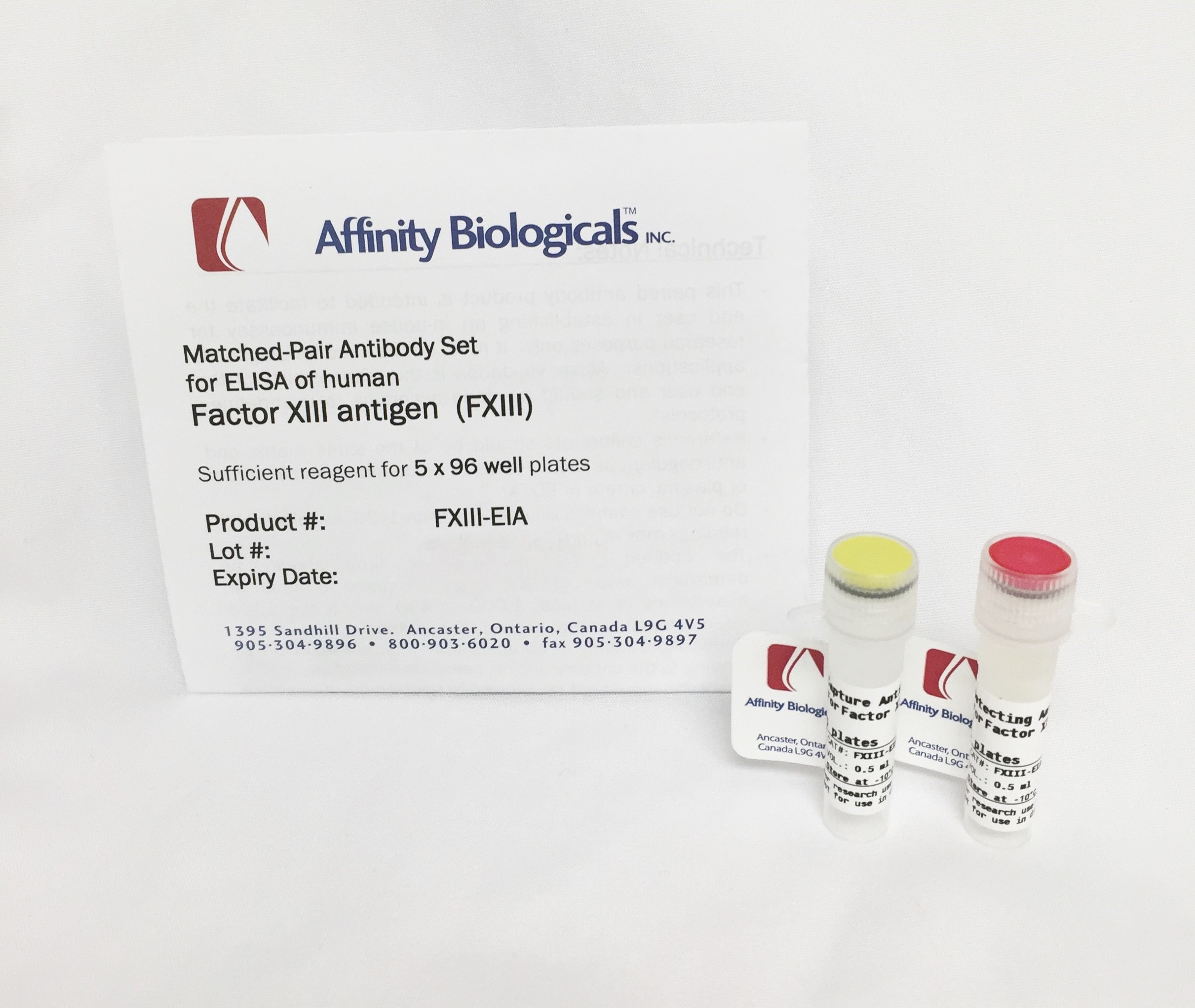
Factor XIII Paired Antibody Set
Paired Antibody SetsAffinity’s Factor XIII Paired Antibody Set consists of matched capture and detecting antibodies that have been titrated and optimized for use in sandwich style ELISA assays. The product as provided contains sufficient capture and detecting antibodies for five full 96-well microplates and contains a detailed protocol sheet containing directions for use, recipes for solutions and sources for additional materials required. This Factor XIII (FXIII or fibrin stabilizing factor) Paired Antibody Set is intended to facilitate the end user in establishing an “in-house” immunoassay for research purposes only and must not be used for diagnostic applications. Assay validation is the responsibility of the end user.
Supplied Materials:
- Capture Antibody (FXIII-EIA-C): One yellow-capped vial containing 0.5 mL of polyclonal affinity purified anti-FXIII Subunit A antibody for coating plates
- Detecting Antibody (FXIII-EIA-D): One red-capped tube containing 0.5 mL of peroxidase conjugated polyclonal anti-Factor XIII antibody for detection of captured Factor XIII
Specifications
Storage and Shelf Life
Storing
between -10°C and -20°C
Type
Frozen
Expiration
Please inquire
Available Formats
| Cat # | Format |
|---|---|
| FXIII-EIA |
1 x 0.5mL Capture Antibody (FXIII-EIA-C) 1 x 0.5 mL Detecting Antibody (FXIII-EIA-D) |

